Our Team

Kristen Perron
I am a fourth-year materials engineering student originally from Austin, TX. I am interested in the electronic device industry which relates to our project on polymers for electronics applications. I am also involved with the Society of Women Engineers and the MATE Honor Society, ASMu.
Kristin Villanueva
I am a fourth-year materials engineering undergraduate with a passion for electronic materials and devices. Outside of the classroom, I dedicate my time to the Society of Women Engineers and the Club Tennis team. I will continue my education as a graduate student in the Cal Poly M.S. Polymers and Coatings Science program.
Connect with us on LinkedIn!
Acknowledgements
Thank you to our project advisors, Dr. Harding and Dr. Zhang. Your support made this project possible.
We would also like to thank Tom Featherstone, Dr. Scott, and Adam Cronin from the Chemistry Department for all their help and advice on this project.
Our Project's Digital Poster
Project Background
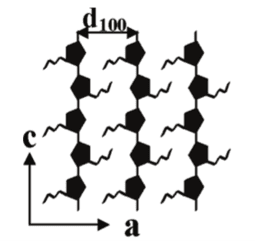
Conductive polymers are becoming a more popular researched field for the consumer electronic market, specifically for applications in flexible electronics such as organic LEDs, solar cells, and field-effect transistors. Poly(3-butylthiophene) (P3BT) is an attractive conductive polymer because in its transcrystalline form, chains of P3BT align and stack on each other to promote conductivity. Producing oriented transcrystals is important, because along the conjugated backbones (c-axis), the charge carrier mobility can be 2-3 orders of magnitude higher than between the side chains (a-axis).
Project Scope
1. Characterize the morphology and conductivity of P3BT transcrystals grown by solvent evaporation method
2. Devise a controlled and reproducible method to grow P3BT transcrystals by controlling vapor pressure of the system
Synthesis
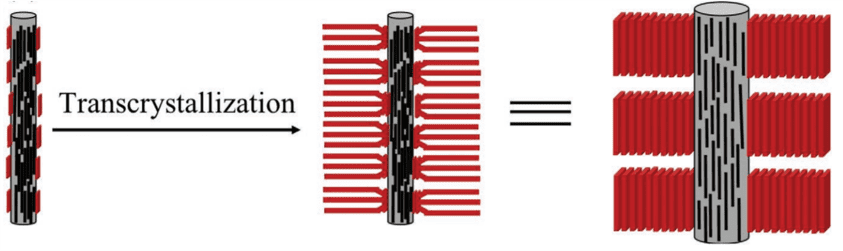
One method to grow transcrystals is the solvent evaporation method using carbon nanotubes as a heterogenous nucleation site. In theory, the transcrystals grow outwardly from the carbon nanotube interface in a columnar fashion, where the conjugated backbone is oriented parallel to the CNT fiber. We selected the solvent evaporation method because it has low fabrication cost and simple processing.
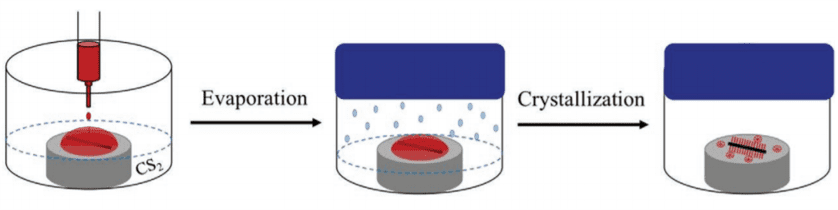
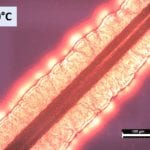
Controlled solvent evaporation starts with preparing a glass substrate with a carbon nanotube and drop-casting P3BT solution on the substrate. We used a solution of carbon disulfide, tri-chlorobenzene, and P3BT. The substrate is then placed and sealed in a jar with carbon disulfide and held at temperature for 24 hours. The carbon disulfide evaporates, and the carbon nanotube acts as a heterogeneous nucleation site for transcrystallization. The solvent evaporation method was used to run a series of trials at 5 different temperatures, 24, 26, 28, 30, and 32⁰C.

This project was advised by Dr. Zhang in the Chemistry and Biochemistry Department and Dr. Harding in the Materials Engineering Department.
Characterization
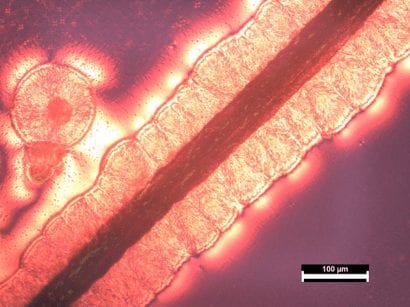
POlarized optical microscopy
Images were taken using polarized optical microscopy (POM). The bright red/orange regions show our polymer growth. Spherulite formation ranging from 50-200um in diameter was observed at 24, 26, 28, and 32⁰C. In spherulitic form, the conjugated backbone is not aligned and therefore conductivity properties cannot be controlled. At 30⁰C, however, transcrystal growth is observed, which is shown by the polymer chains radiating outward in a columnar fashion.
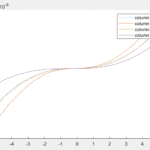
Length measurements
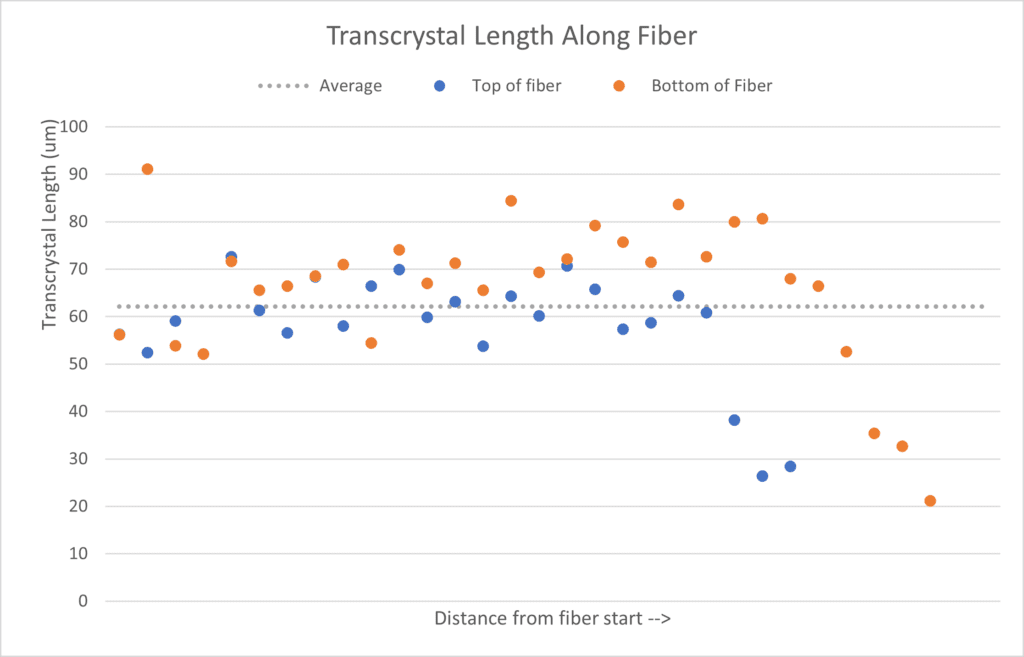
The software, ImageJ, was used to measure transcrystal length of the 30⁰C. The plot shows the length of the transcrystals along the top and bottom of the fiber. A random distribution of transcrystal lengths along the fiber was observed, ranging from 20-90 um. The average transcrystal length was 61.6 microns with a standard deviation of 25.9. Literature values report average lengths ranging from 100-900um, while other reports show average lengths from 1-50um. Since the data ranges greatly, we concluded that the growth process should be adjusted in order to produce transcrystals more consistently.
AFM Results
Morphology and conductivity
Using AFM in tapping mode, we were able to produce a topographical image of the transcrystals. The lamellae of the transcrystals grow perpendicular to long axis of the CNT fiber in a 1D nanowire-like orientation. Using silver paint as an electrode on one end of the CNT fiber and a conductive AFM tip, IV curves were taken at incremental distances from the fiber.
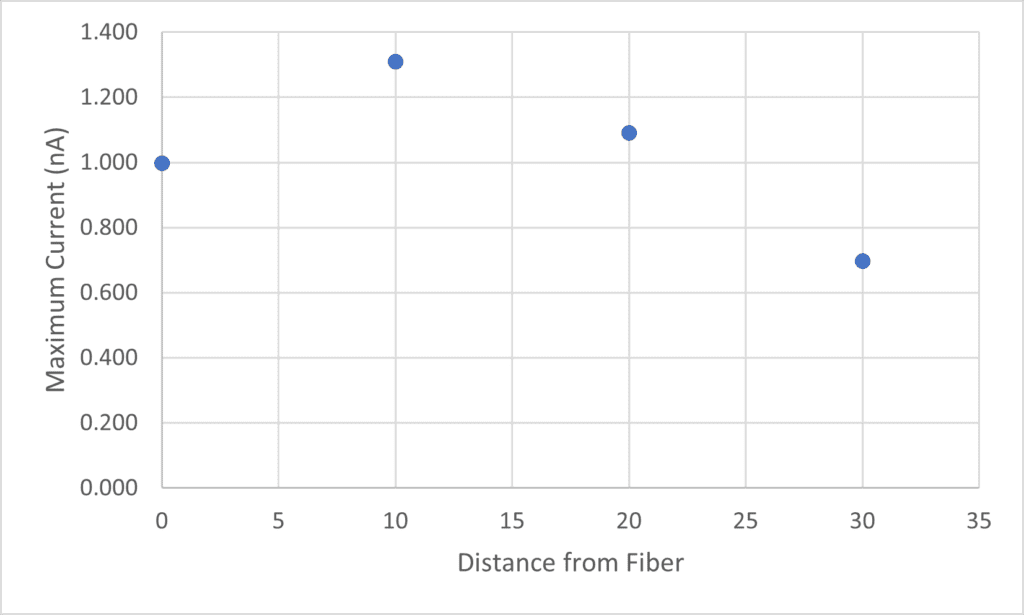
This graph shows the maximum current from each IV curve (each distance from the fiber). At 10um away, we see a max current of 1.3nA, at 20um away, we see a max current of 1.1nA, and at 30um away, we see max current of 0.7nA. This shows a trend we would expect that with increasing distance from the fiber, the transcrystals show decreasing current. The CNT fiber showed a lower current than nearby on the transcrystal which could be due to a tear in the fiber or probing on the edge of the fiber.
New Process Design
Controlled vapor pressure system
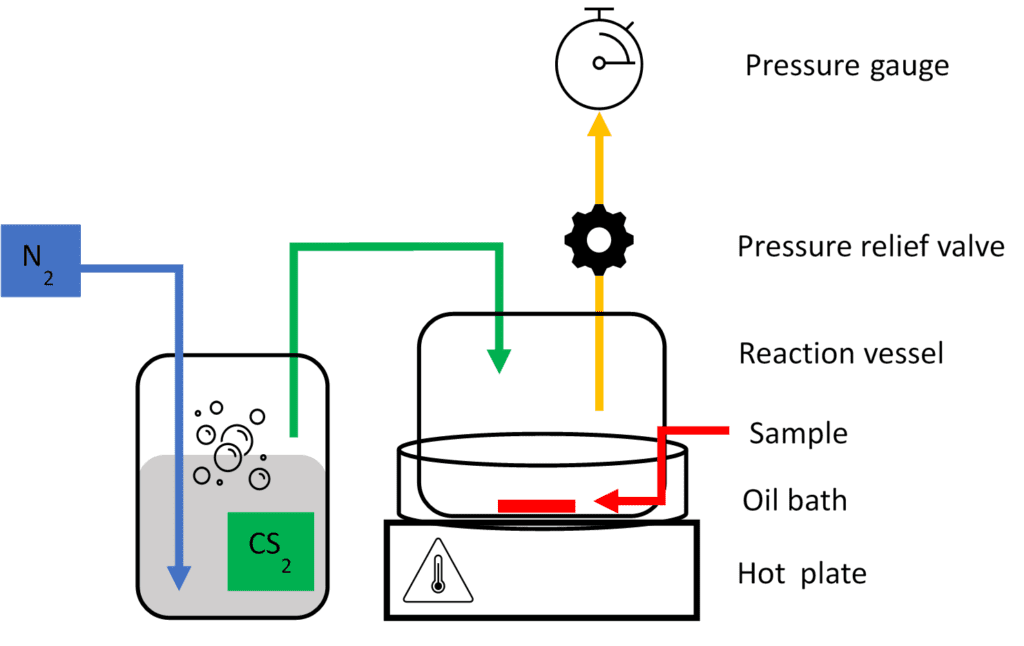
Nitrogen gas bubbles the liquid carbon disulfide and pumps carbon disulfide vapor into the reaction vessel, which contains the glass slide sample. A pressure relief valve and pressure gauge maintain a constant regulated vapor pressure within the reaction vessel. This process maintains control over the vapor pressure in order to produce transcrystals more consistently.
Conclusions
1. Transcrystals grown by solvent evaporation showed an average length of 61.6 μm
2. A new growth method for reproducible transcrystal growth was devised
3. A method to measure transcrystal conductivity was developed and implemented
4. P3BT transcrystals showed decreasing current with increasing distance from the CNT fiber